When it comes to the process of our joints healing, one factor is crucial: time. It takes time for the affected joints to be stabilised using non-invasive techniques.
With its broad range of supports and orthoses, L&R supports this healing process, offering the right product for each individual case. These products stand out due to the modern materials they contain, their ease of use, their wearing comfort, and their streamlined sizing systems. Countless exciting innovations have been seen in this field in recent months, greatly enriching the market.
So, how does a new product actually become a reality?
We have set off on an adventure with L&R into the world of supports and orthoses: the company’s Research & Development team has been working closely together with Global Portfolio Management. Whilst R&D has historically been based in the French city of St. Genest for historical reasons, Global Portfolio Management is located in a German city, Rengsdorf. Despite cultural differences, the team of over 10 people from different professions and backgrounds has come together: engineers, medical experts and product specialists, all collaborating in one team. The group holds regular meetings to ensure continuous collaboration to find new product innovations and modifications, striving to meet patients’ exacting needs down to the very last detail.
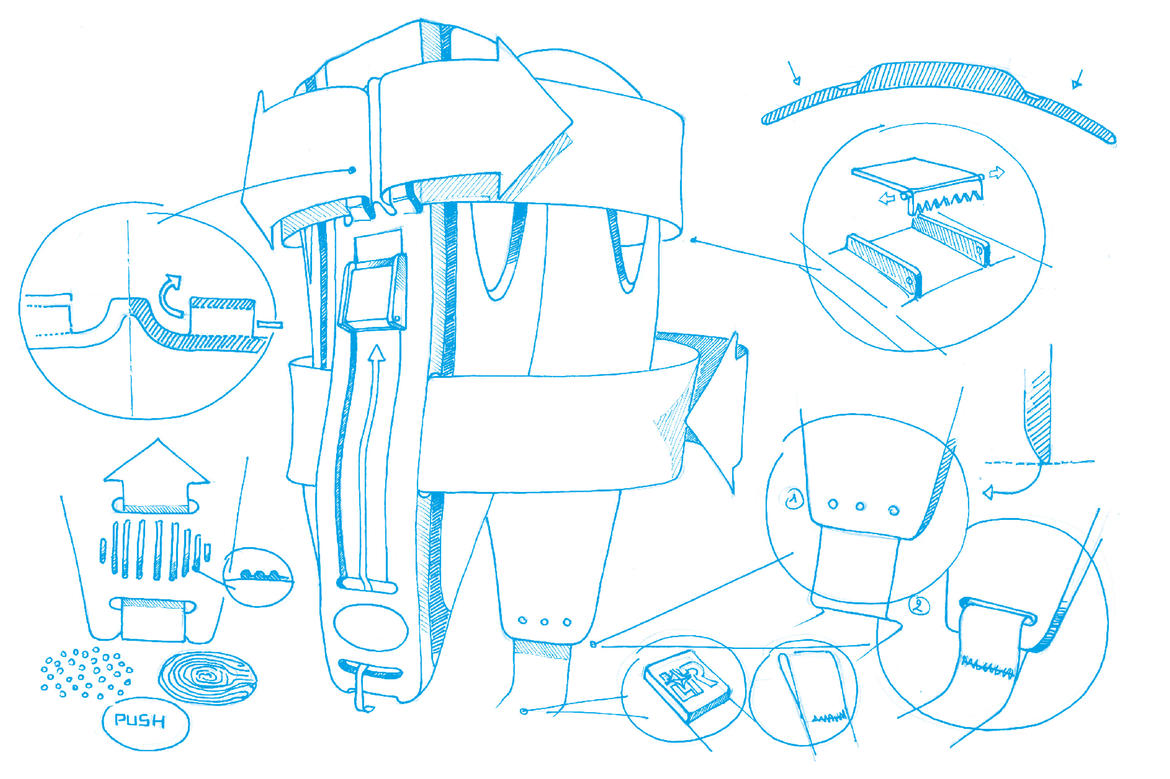
At the start of a development process of this type, the team faces the question of what products the market is showing a demand for. And the key factor here for L&R’s solution providers? The indication, i.e. the problem the patient has presented with, for which a solution may need to be found.Once the demand from the market has been identified, and the problem recognised, the development team gets to work. Is there already a product in the portfolio which would be improved through modification? Or would it make more sense to start from scratch? A unique selling point (USP), which sets the product apart as unique and better than products from the competition, is also developed as part of this process. That’s how L&R came to develop the flexible side shells on the new Malleo Control Comfort, for example, ensuring that they can be adapted to each individual patient’s leg shape to guarantee maximum comfort. It can also be of significant benefit if a product can be worn on either the left or the right foot, like the new Cellacare Rhizo Classic; or if a product’s size can be adjusted to suit each individual, as is the case for the Cellacare Clavicula Classic.
All of these considerations lead to a product idea, which is not simply recorded on paper, but is passed on to colleagues from Research & Development who create a digital model from the plans.
Further details are worked out in parallel: What material should the new product be made from? Which details need to be taken into account during manufacture? The next step involves creating and evaluating different prototypes, and a 3D digital illustration is developed.
Various different employees are involved in the task of checking the prototypes and making further improvements where necessary. Is everything working as originally planned? Does the product meet the technical requirements? Does it comply with all the specifications as per the legal requirements and the health insurance providers’ requirements? When both Research & Development and Global Portfolio Management, as well as the team of medical experts are all satisfied, a 3D printer is used to create the first model of the new product, which will then be used as the basis for manufacture.
Before a new product can be certified, comprehensive user and usability tests must be carried out, whereby product claims in terms of the product characteristics can be scientifically proven. After several months have passed, and the product has been examined from every possible angle, a new orthosis is now developed and can start supporting patients in their treatment.
But the work does not stop there, not even close. Once the product is launched to market, L&R starts a continuous exchange with customers and patients, taking feedback on board and once again collating ideas and new directions for future optimisation.
Rhizocast Classic